20.109(F16):Purify active materials (Day2)
Contents
Introduction
Biomineralization is a process by which living organisms generate minerals that are often used to harden soft tissues. In our work for module 3 we are using M13 as a template for the biomineralization of phosphate to be used in your Li(FePO4) cathode.
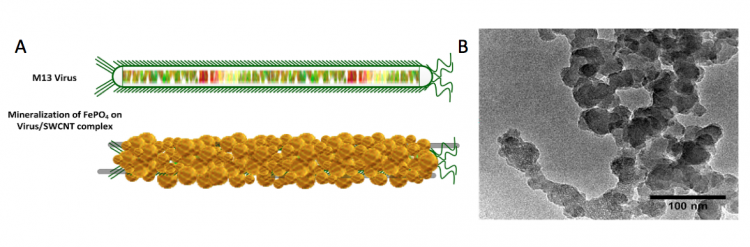
The use of M13 as a 'scaffold' for this reaction is also termed biotemplating. Biotemplating utilizes biological materials to fabricate and/or organize unique nanomaterial structures. A variety of benefits are associated with biotemplated synthesis methods over traditional synthesis methods. From the fabrication standpoint, biotemplating can reduce the environmental impact of materials synthesis. Biotemplating has been shown to enable synthesis of materials at lower temperatures and pressures as well as using milder reagents than traditional synthesis methods. From the standpoint of organization, biotemplating can achieve unique structures over broad length scales. These structures are either unattainable or significantly more difficult to create without biotemplating.
Protocols
Part 1: Visit from BE Communication Lab
Today Dr. Sean Clarke from the BE Communication Lab will join us to provide some tips for your research proposal presentation.
Part 2: Demo of phage biomineralization reaction
Because the initial step of the biomineralization process is time-sensitive, the instructors quenched your reactions after ~12 h by adding 50 mL of 10 mM Na:PO4. In this step, the phosphate precipitated the Fe(III) onto the p8 proteins of the M13 phage coat to generate the nanowires that will be used to construct the cathode of your battery.
So you can see this reaction, we also prepared a small sample as a demo. This morning, the biomineralization reaction was initiated by mixing 500 μL of phage (at 1.7 x 1013 pfu/mL) with 500 μL of a 10 mM (NH4)2Fe(SO4)2 solution in a 2 mL eppendorf tube, and incubated for 6 h.
- Quench the biomineralization reaction by adding 500 μL of a 10 mM Na:PO4 solution.
- What do you observe?
Part 3: Collect active material (nanowires)
In your flask are the Fe(III)-phage-AuNP biomineralized nanowires that will serve as the active material in your battery cathode. Today you will isolate and wash this material.
- Retrieve your flask with your Fe(III)-phage-AuNP nanowires from the front bench.
- Gather the nanowires that adhered to the side of your flask.
- Use a P1000 pipet to 'waterfall' the culture liquid from your flask down the sides of the flask.
- Divide the liquid in your flask equally between three 50 mL conical tubes.
- Be sure to clearly label your tubes with a marker as stickers will come off in the centrifuge.
- Centrifuge at 3,250 rpm for 20 minutes.
- Decant the supernatant in the sink.
- Carefully remove any residual liquid with your P1000.
- Resuspend your pellet in diH2O.
- Add 2 mL of diH2O and mix by pipetting up and down ~10 times.
- Continue adding 1-2 mL aliquots of diH2O and pippetting to mix until you reach a final volume of 4 mL in each tube.
- Transfer the resuspended nanowires from all three 50 mL conicals to a single 15 mL conical.
- Be sure to clearly label your tube.
- Centrifuge at 3,250 rpm for 20 minutes.
- Decant the supernatant in the sink.
- Resuspend your pellet in 10 mL diH2O.
Part 4: Prepare TEM grid with nanowires
Each group should prepare two TEM grids: one of the straight nanowire solution, and one of a 1:10 dilution. You will visualize your nanowires on M3D4 with Prof. Belcher and George Sun.
- Vortex the nanowire pool for 1 minute at least and immediately remove 10 μL of the nanowire suspension to place on the silver/black side (not the copper-looking side) of the TEM grid that you have balanced in the specialized tweezers. Treat the grid with care and use the tweezers only on the edge to minimize damaging the delicate mesh.
- Prepare a 1:10 dilution of nanowire pool by mixing and vortexing 5 μL of the original solution with 45 μL diH2O. Dispense 10 μL of this dilution onto a second TEM grid.
- Allow the nanowires to settle onto the grid undisturbed for 20 minutes.
- Remove the droplets from the grids by touching the edges with a Kimwipe, thereby wicking the solution off the grid.
- Wash the grids by adding 10 μL of sterile water onto each grid.
- Allow the grids to sit with water for 3 minutes and then wick dry.
- Wash the grids by adding 10 μL of 100% EtOH onto each grid.
- Allow the grids to sit with EtOH for 3 minutes and then wick dry, if it hasn't air dried.
- Place the grids into the TEM grid holder for safe transport to the TEM facility (13-1012).
Part 5: Dry active material (nanowires)
To further prepare your nanowires for use in your battery, this active material will be dried at high temperature to remove all liquid.
- Centrifuge your resuspended nanowires from Part 3 at 3,250 rpm for 20 minutes.
- Decant the supernatant in the sink.
- Carefully remove any residual liquid with your P200.
- Cover your tube with aluminum foil and punch a small hole in the top.
- Bring your tube tube to the front bench.
Your tubes will be transported to the Belcher Laboratory and will be placed in a vacuum drying oven at 80°C.
Reagents list
- Copper film TEM grids (400 mesh) from Electron Microscopy Sciences
- 100% ethanol
Next day: Cathode construction
Previous day: Growth of phage materials
