Difference between revisions of "Spring 11:Chemotaxis Assay"
| Line 55: | Line 55: | ||
=== Basic Design === | === Basic Design === | ||
| − | The fundamental principle behind this system is that changes in pressures causes fluid flow and when the pressure equalize to the same 'ground' pressure, fluid flow should stop. By this approach one is able to easily control fluid flow of the entire channel and the flow rates into inlets A and B to control the width of the chemoeffector band, dependent on the ratio of | + | The fundamental principle behind this system is that changes in pressures causes fluid flow and when the pressure equalize to the same 'ground' pressure, fluid flow should stop. By this approach one is able to easily control fluid flow of the entire channel and the flow rates into inlets A and B to control the width of the chemoeffector band, dependent on the ratio of P_{A} and P_{B}. Solenoid actuators, controlled by LabView, to switch from the 'flow' pressure state to the 'stopped' state. |
[[File:Systemized_Chemotaxis_Assay_Presentation_1.jpg|center|thumb|600px|'''Figure 4: Pressure states of fluid flow]] | [[File:Systemized_Chemotaxis_Assay_Presentation_1.jpg|center|thumb|600px|'''Figure 4: Pressure states of fluid flow]] | ||
| Line 62: | Line 62: | ||
=== Circuit Element Design === | === Circuit Element Design === | ||
| + | In order to understand the system, I modeled it as a lumped circuit elements model ([http://holbert.faculty.asu.edu/analogy.html Interdisciplinary Electrical Analogies]). All of the pressure potentials are measured relative to atmospheric (atm = 0 psi) | ||
| + | |||
| + | * Flow/Current: Volume (fluid) flow = <math> G </math> [<math>m^{3}\cdot s^{-1}</math>] | ||
| + | * Potential: Pressure drop, P [psi] | ||
| + | * Integrating Element: Inertance = <math> M </math>, <math>G = \frac{1}{M} \int \rho {\mathrm{d} t} </math> | ||
| + | ** e.g., for pipe: <math> M = \rho*\frac{L}{A} </math> | ||
| + | * Fluid resistance: <math> R </math> [<math>m^3*s^{-1}\cdot psi^{-1}</math>], <math>R = G/P </math> | ||
| + | * Fluid capacitance: <math> C </math> [<math>m^{3}\cdot psi^{-1}</math>], <math>G = C*\frac{\mathrm{d} P}{\mathrm{d} t} </math> | ||
| + | |||
| + | |||
| + | ====Design 1==== | ||
| + | |||
| + | ====Design 2==== | ||
| + | |||
| + | ====Design 3==== | ||
Revision as of 05:53, 17 May 2011
Contents
Introduction
For my project I designed and developed an automated microfluidic system using Seymour et al’s microinjector microfluidic device to provide a high through-put method of acquiring bacterial chemotaxis parameters. The microinjector device was chosen for its simple design, diverse application to both fluid-flow and flow-free approaches, and for its ability to measure individual cell kinetics. The development of a faster and systematic device is especially useful in the research of marine microbes’ response to settling dissolved organic matter.
Objective
Further develop Stocker’s microfluidic system that will allow researchers to run chemotaxis assays more efficiently for high though-put assays. The main improvement to Stocker's system will be the use of pneumatic pressures instead of syringe fluid pressures to control fluid flow to set up the chemoatractant and microorganism bands. The pneumatic pressure will be computer controlled to to allow precise and reproducible experiments as well as increased experimental throughput.
Spring 20.345 Project Assignments
- [add files here, ask steve]
Readings and Resources
- [add files and links here, ask steve]
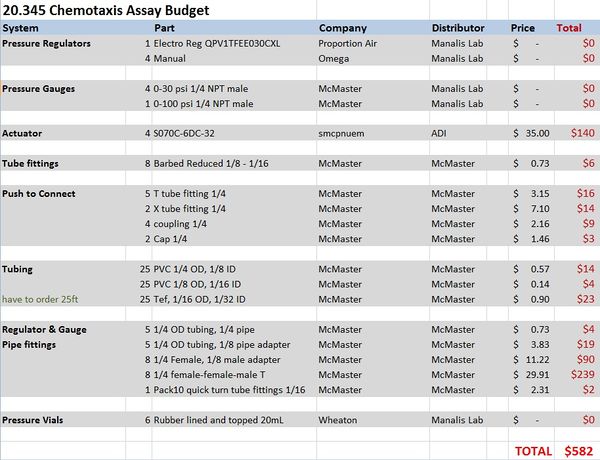
Materials and budget
Microinjector Microfluidic Device
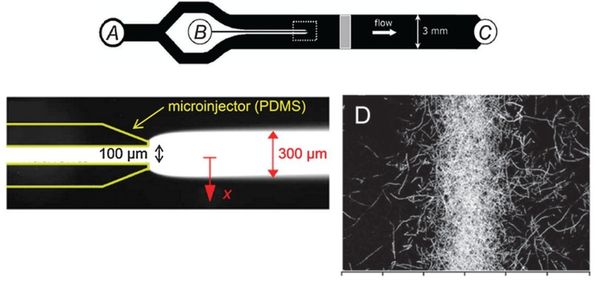
Flow-free, or ‘stopped-flow’, chemotaxis assays allow for the analysis of individual bacterial movement in the absence of shear forces and relies on natural diffusion to generate a gradient. In this approach, fluid flow is only used to set up an initial gradient, but is stopped, allowing the gradient to evolve by diffusion alone. These devices produce unsteady gradients that are advantageous in representing environmental conditions of nutrients especially in marine bacteria and in characterizing bacterial responses to wide range of concentration and gradients within a single experiement. The bacterial distribution is measured by recording the kinetic paths of individual bacteria using videomicroscopy. An example of this approach is Seymour et al’s microinjector microfluidic device
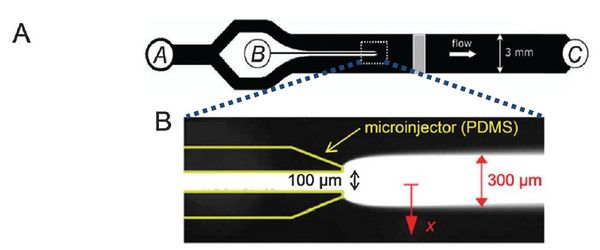
Dimmension
- Microinjector Channel
- width: 100μm
- depth: 50μm
- length:
- Main Channel
- width: 3mm
- depth: 50μm
- length:
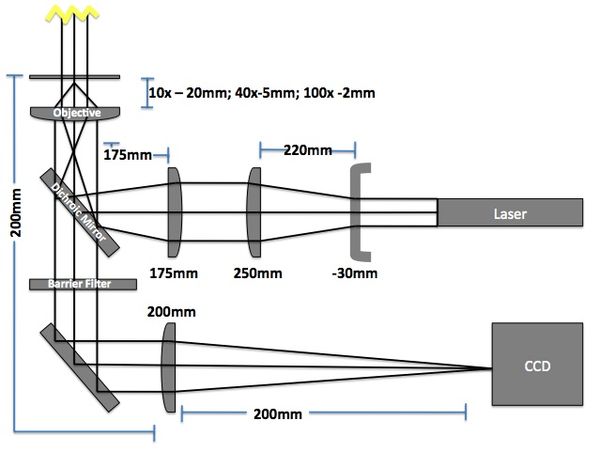
Microscope
The imaging microscope will be a brightfield and fluorescent microscope as shown in Fig 3. A CCD camera will acquire images at a given rate and the kinetic paths will be tracked using particle tracking algorithms.
- Camera (Allied Manta G032B): 7.4μm sqr pixels, 656 x 492 pixels
- Field of View: 484μm x 366μm (10x objective w/200mm lens => 10x magnification)
- Red LED illumination
Pneumatic System Design
The pneumatic system is composed of manual and electro forward pressure regulators, solenoid actuators, pneumatic tubing, and circuitry to
Basic Design
The fundamental principle behind this system is that changes in pressures causes fluid flow and when the pressure equalize to the same 'ground' pressure, fluid flow should stop. By this approach one is able to easily control fluid flow of the entire channel and the flow rates into inlets A and B to control the width of the chemoeffector band, dependent on the ratio of P_{A} and P_{B}. Solenoid actuators, controlled by LabView, to switch from the 'flow' pressure state to the 'stopped' state.
Circuit Element Design
In order to understand the system, I modeled it as a lumped circuit elements model (Interdisciplinary Electrical Analogies). All of the pressure potentials are measured relative to atmospheric (atm = 0 psi)
- Flow/Current: Volume (fluid) flow = $ G $ [$ m^{3}\cdot s^{-1} $]
- Potential: Pressure drop, P [psi]
- Integrating Element: Inertance = $ M $, $ G = \frac{1}{M} \int \rho {\mathrm{d} t} $
- e.g., for pipe: $ M = \rho*\frac{L}{A} $
- Fluid resistance: $ R $ [$ m^3*s^{-1}\cdot psi^{-1} $], $ R = G/P $
- Fluid capacitance: $ C $ [$ m^{3}\cdot psi^{-1} $], $ G = C*\frac{\mathrm{d} P}{\mathrm{d} t} $
Design 1
Design 2
Design 3
Experiment 1:
- By Emmanuel Quiroz