20.109(F15):Battery assembly and testing (Day5)
Contents
Introduction
Today you will assess the data to determine how well your battery performed. Previously, you prepared a battery electrode, specifically a cathode, from Fe(III)-phage nanowires that you combined with carbon. The redox properties of this active material determine the operating voltage, while other properties influence the capacity (how long the battery 'lasts' under a given current load) and rate capability (how quickly the battery is discharged or charged). Capacity and rate capability can be improved by making materials very small or by incorporating conductive metals into the matrix into the material. The goal of your research is to assess how the amount of phage used in biomineralization affects capacity.
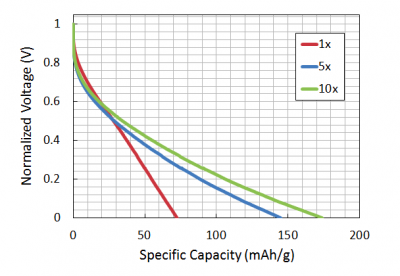
The theoretical capacity of a battery is the quantity of electricity involved for the electro-chemical reaction within the battery. For our Fe(III)-phage batteries, the theoretical capacity is 178 mA*h/g, a value based on the cathode material - FePO4. Using this information, we can calculate the "loading factor" for the Galvanostat to be 17.8 mA/g (theoretical capacity divided by the time used to charge/discharge). This is the amount of current to use for a 10 hour discharge. The discharge time is selected by the researcher. Slower discharge times are better as diffusion of the solvents is increased making it more likely that your battery will reach the theoretical capacity.
To calculate the current needed to fully charge or discharge your battery, you need to account for the mass of your cathode. The mass of every electrode will be a little different, so the charge/discharge rates will be slightly different. But as an example, if your battery cathode is precisely 1 mg of active material, then to discharge it in 10 hours we'd have to apply a negative current of -17.8 μA.
It is important to note that your cathode consists of more than just the active material. Remember that when you prepared your cathode material you used 70% Fe(III)-phage nanowires, 25% Super P, and 5% PTFE...in addition your Fe(III)-phage nanowires are ~10% phage. To account for this, you need to adjust your cathode weight by multiplying by 0.63. Be sure to know how this value was determined!
Protocols
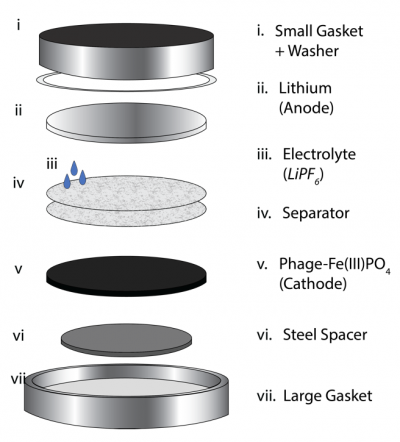
Part 1: Battery assembly
Because the battery testing step requires several hours, George assembled and tested your batteries last week while you were away. Thank you, George! Today you will see a demo of the battery assembly procedure. For your notes, the steps are provided below.
- blah
Part 2: Battery testing
Reagents list
Next day: Research proposal presentations
Previous day: TEM