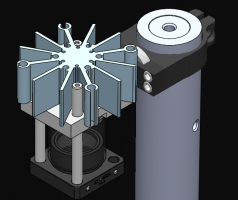
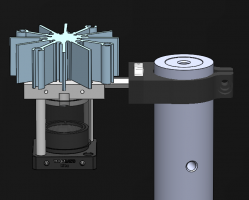
Assignment 1, Part 3: Building your transillumination microscope
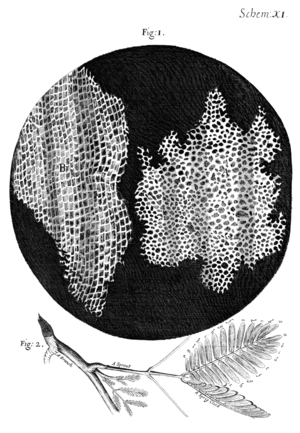
I took a good clear piece of Cork, and with a Pen-knife sharpen'd as keen as a Razor, I cut a piece of it off, and thereby left the surface of it exceeding smooth, then examining it very diligently with a Microscope, me thought I could perceive it to appear a little porous; but I could not so plainly distinguish them, as to be sure that they were pores, much less what Figure they were of: But judging from the lightness and yielding quality of the Cork, that certainly the texture could not be so curious, but that possibly, if I could use some further diligence, I might find it to be discernable with a Microscope, I with the same sharp Penknife, cut off from the former smooth surface an exceeding thin piece of it, and placing it on a black object Plate, because it was it self a white body, and casting the light on it with a deep plano-convex Glass, I could exceeding plainly perceive it to be all perforated and porous, much like a Honey-comb, but that the pores of it were not regular; yet it was not unlike a Honey-comb in these particulars.
I told several lines of these pores, and found that there were usually about threescore of these small Cells placed end-ways in the eighteenth part of an Inch in length, whence I concluded there must be neer eleven hundred of them, or somewhat more then a thousand in the length of an Inch, and therefore in a square Inch above a Million, or 1166400. and in a Cubick Inch, above twelve hundred Millions, or 1259712000. a thing almost incredible, did not our Microscope assure us of it by ocular demonstration.
— Robert Hooke from Micrographia: or Some Physiological Descriptions of Minute Bodies made by Magnifying Glasses with Observations and Inquiries Thereupon (1665)[1]
Background
In the remaining parts of this assignment, you will build a compound microscope, determine its magnification, and attempt to measure the size of microscopic objects. The instrument you create will have a great deal in common with the microscope Robert Hooke built in the mid-1660s. Hooke meticulously documented his microscopic observations and published them in a popular volume called Micrographia in 1665. The measurements you make in part 1 will call to mind Hooke's early quantification of the size of plant cells (see quote at top of page). You will grapple with many of the same challenges Hooke faced: resolution, contrast, field of view, optical aberrations, and obscurity of thick samples. (To overcome the thick sample problem, Hooke used a very sharp knife to cut an "exceeding thin" slice of cork — a technique still in everyday use.)
Hooke spent countless hours hand drawing the breathtaking illustrations for Micrographia. A CMOS camera in the image plane of your microscope will provide a huge advantage. You will be able to record micrographs nearly as spectacular as Hooke's in a fraction of a second and with far less skill. (As a young man, Hooke apprenticed as a painter. The guy could draw.)
Specimens in Assignment 1 will be illuminated by an LED that shines light through the sample plane. The illumination will show up as a bright background in your images. The unsurprising name of this method is: transilluminated, bright field microscopy. Transillumination works well for samples that absorb or scatter a lot of light. Most biological samples have low contrast when imaged this way. Despite the limitations of bright field microscopy, many important discoveries were made with this simple method. Hooke was an early discoverer of plant cells, but he was mostly interested in how the cell structure of his cork sample explained the material's unique mechanical properties. He soon trained his microscope on other things (like glass canes, a bloodsucking louse, and feathers).
Likely inspired by Micrographia, a Dutch draper named Anton van Leeuwenhoek honed his lens-making skills and developed his own microscope. Van Leeuwenhoek was intensely interested in the tiny creatures he dubbed "animalcules" that he observed in water, blood, semen, and other specimens. Looking at samples of plaque from his own mouth, van Leeuwenhoek recorded: "I then most always saw, with great wonder, that in the said matter there were many very little living animalcules, very prettily a-moving. The biggest sort. . . had a very strong and swift motion, and shot through the water (or spittle) like a pike does through the water. Looking at the second sort. . . oft-times spun round like a top. . . and these were far more in number." (Sadly, the colorful term "animalcule" did not have as much staying power as "cell.") Van Leeuwenhoek discovered bacteria, protozoa, spermatozoa, rotifers, Hydra, Volvox, and parthenogenesis in aphids. He was truly the first microbiologist.
Perhaps the most remarkable discovery ever made with nothing but a simple light microscope was genetic transposition. Barbara McClintock was a talented microscopist who developed a technique that enabled her to distinguish individual chromosomes in Zea mays (corn) plant cells. One important element of her method was that she prepared her samples by squashing them instead of cutting thin slices as Hooke did 300 years earlier. Squashing tended to preserve the chromosomal structure better than slicing. She observed genetic transposition through an optical microscope in 1944, nearly 10 years before the chemical structure of DNA was deciphered. Several decades elapsed before molecular techniques sufficiently sophisticated to confirm her discovery were developed.[2] McClintock was awarded the Nobel Prize in Physiology or Medicine in 1983 for her discovery.
Overview
Microscope design
There is more than one way to arrange the optics in a microscope. The particular design you will use in this lab calls for two lenses: an objective lens with focal length $ f_{objective} $ and a tube lens with focal length $ f_{tube} $. As shown in the diagram below, the sample sits at the front focus of the objective lens. The distance between the objective and the tube lens is equal to the sum of their focal lengths. An image with magnification $ \frac{f_{tube}}{f_{objective}} $ is formed at the back focal plane of the tube lens. This is the same arrangement of lenses that you solved for in Assignment 1, Part 1: Pre-lab questions, number 5; it's called a 4f system because it is the sum of 4 focal lengths $ 2\times f_{objective} $ + $ 2 \times f_{tube} $. Yes, it's a pretty bad name.
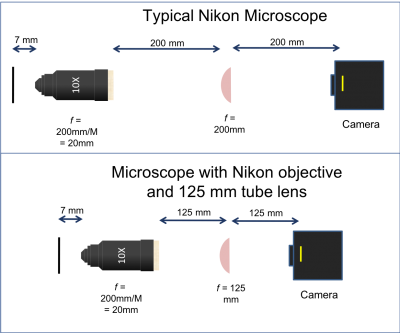
Microscope objectives generally have their magnification printed on the side, not their focal length. A simple formula converts magnification to focal length: $ f_{objective}=\frac{ETL}{M} $, where $ ETL $ is the effective tube length and $ M $ is the magnification. As a consequence, a microscope built with a different tube lens will have a different magnification than what's printed on the objective.
There are three objective lenses available in the lab: a 10×, a 40×, and a 100×. All of these are designed to use a 200 mm tube lens to form an image on the camera. To optimize the signal to noise of our microscope, we are going to use a 125 mm tube lens instead of Nikon's conventional 200 mm lens.
- Working distance (WD) is the distance between the front objective lens surface and the cover slip, and so it is also approximately the distance to the front focal plane. In order to focus an image at the back focal plane of the tube lens, i.e., on the CMOS array, the sample plane must coincide with the front focal plane in a 4f microscope arrangement. The stage is added to hold the sample in this plane.
- The 100× objective is designed to be used with immersion oil. When using the 100× objective, place a drop of oil directly on the tip on the objective. Bring the drop in contact with the slide cover glass. After use, clean off the remaining oil by wicking it away with lens paper or a Kim-wipe. Do not put samples away dirty.
- Note that the back focal plane (BFP) of the objective does not necessarily coincide with the rear of the objective housing. In fact, for the Nikon 40x objective the BFP is close to the blue ring. You will find its actual location when aligning the fluorescence illumination path in Assignment 2. The 200 mm distance labeled between the back of the objective housing and the tube lens is a recommendation from Nikon to enable optimal imaging. For details on the importance and origin of this distance please ask an instructor.
Please see the Nikon Introduction to Microscope Objectives at their excellent MicroscopyU website.
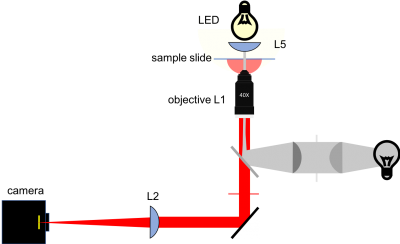
The figure below shows a more detailed block diagram of the instrument you are going to build. By the time you are done, it will be capable of making wicked-cool images using two different kinds of contrast: transilluminated bright field, and epi-illuminated fluorescence. You will only use transillumination in this assignment. We'll add all the stuff for epifluorescence in the next one. The components shown in gray are for implementing epifluorescence contrast. You can leave most of the fluorescence components out for now; you will put them in when you get to Assignment 2. But it happens that two of the components for epifluorescence share the same optical path with transillumination — the dichroic mirror and the emission filter. If you make accommodations for these two parts while you are building this week, you won't have to take your whole microscope apart next week when you add fluorescence. Don't fret. The instructions will tell you what to do.
An example microscope made by the instructors will be available for you to examine. Look it over before you start screwing parts together to get an idea of what you are getting yourself into. The sections below introduce some of the key components the microscope is made out of. Manufacturers such as Thorlabs, Newport, and Edmund Optics sell optical components and prototyping hardware. The majority of parts in the '309 Lab come from Thorlabs, although all three of these manufacturers are represented.
Optical components and equipment
Lenses
Plano-convex spherical lenses are available with focal lengths of 20, 25, 35, 50, 75, 100, 125, 150, 175, 200 and 250 mm. Plano-concave lenses with focal lengths of -30, -50, and -75 mm are also available. It is best to mount most optics in short (e.g. 0.5") lens tubes. It is acceptable to mount a lens between the end of a tube and a tube ring or between two tube rings. In most cases, the convex side of the lens faces toward the collimated beam; the planar side goes toward the convergent rays.
- Tip: Verify all optics before you use them by determining the focal length with a ruler. You can use the lens measuring station. Alternatively, you can use the ceiling fluorescent lamps as a light source and measure the exact distance between the lens(es) assessed and the lamp's image. Can you imagine a simple rig to evaluate negative focal lengths (of plano-concave lenses for instance)?
- Tip: As you install lenses into your microscope, put a piece of tape on the lens tube showing focal length and orientation. This will help you both during construction and put-away. Save the lens storage boxes and return components to the correct boxes when you are done.
- Handle lenses only by the edges. If a lens is dirty, first remove grit with a blast of clean air or CO2. Clean the lens by wiping with a folded piece of lens paper wetted with a drop of methanol. (Do not touch the part of the tissue you use for cleaning with your fingers.) In some cases, it may be helpful to hold the folded lens tissue in a hemostat. Ask an instructor if you need help.
Screw sizes
Your microscope will be held together with screws. Here's how to identify the correct fastener:
Screw sizes are specified as <diameter>-<thread pitch> x <length> <type>. The diameter specification is confusing. Diameters ¼" and larger are measured in fractional inches, whereas diameters smaller than ¼" are expressed as an integer number that is defined in the Unified Thread Standard. The thread pitch is measured in threads per inch, and the length of the screw is also measured in fractional inch units. So an example screw specification is: ¼-20 x 3/4. Watch this video to see how to use a screw gauge to measure screws. (There is a white, plastic screw gauge located near the screw bins.) The type tells you what kind of head the screw has on it. We mostly use stainless steel socket head cap screws (SHCS) and set screws. If you are unfamiliar with screw types, take a look at the main screw page on the McMaster-Carr website. Notice the useful about ... links on the left side of the page. Click these links for more information about screw sizes and attributes. This link will take you to an awesome chart of SHCS sizes.
Tools
Most of the tools you will use are located in the top drawers of the cabinets under your lab station. Hex keys (also called Allen wrenches) are used to operate SHCSs. Some hex keys have a flat end and others have a ball on the end, called balldrivers. The ball makes it possible to use the driver at an angle to the screw axis, which is very useful in tight spaces. You can get things tighter (and tight things looser) with a flat driver. The spanner wrench is used to tighten and loosed the SM1 retaining rings that hold lenses and other optics into place in the Thorlabs SM1 threaded system.
- Some of the tools located in your station's drawers:
Rigid optical construction
There are several ways to construct an optical systems: rails, posts, cages, tubes, and all manner of confusing, little, metallic bits. The 20.309 example microscope is constructed chiefly using the Thorlabs 30 mm cage system and 1" lens tubes. The point of these systems is to hold the optics securely in their proper positions while also allowing things require adjustment to be moved as needed. The structure of your microscope should be solid. If not, you will find the optics go out of adjustment each time you put the 'scope away. Also, in Assignment 5, you will use your instrument to track nanometer-scale motions of particles inside of cells. If the components of your microscope are jiggling around, you won't be able to get good results on that part of the lab.
The cage system consists of stainless steel cage rods that fit into matching holes in cage plates. There are four set screws in the sides of each cage plate. (There is another similar-looking kind of cage plate with eight set screws. Make sure you use the right kind.) Tightening the set screws fixes the rods in place. The figure below shows how the set screws engage the cage rods to hold them in place. You don't need to take the set screws all of the way out — just make them loose enough so that you can insert the cage rod. When you want the cage plate to stay fixed in the same spot, use the widdy-biddy hex driver to tighten the set screw. It doesn't make sense to try and get the set screws super-tight. You'll just round off the head of the driver, instead. Make the set screws just tight enough. If you need to move the cage plate later on, just back the screw off a half turn.
The best way to mount a ordinary circular optics like lenses or mirrors or filters in a cage is to first put it in a half-inch lens tube. Then screw the tube into a cage plate. That way, you can adjust the position of the optic by sliding the cage plate. (Sliding adjustments are almost always better than rotating ones, which are usually annoying. The optical center of lenses is always a little off, and optics never lie perfectly flat. So the beam path changes when you rotate an optic. Exasperating.)
The most common kind of cage plate has threads that allow you to screw a lens tube into it. The pretty pictures in the Thorlabs catalog always show four rods. Don't fall for it — use only three rods when you build a 30mm cage. Lens tubes can't pass between two 30 mm rods; they get stranded inside the cage. "Ha ha, got you again," snickers the V.P. of Marketing whenever a hapless customer has to take their entire instrument apart in order to clean a lens or change an optic that gets stranded inside a four-rod cage. It's usually best to eliminate one of the bottom rods so you can hang stuff on the top rods. You'll see why later.
Sample stage
A precision Newport X/Y/Z stage[3] with a sample holder mounted on a post, or a Thorlabs Max312D stage, also with a sample holder, is available at each lab station. The Newport stage setup is top-heavy. Avoid accidents by ensuring that the post base is always attached to an optical breadboard or table. Leave the stage at the lab station when you are done with it. For the Thorlabs stages, it is still a good idea to bolt them down so that your area of interest (AOI) stays in your microscope field of view (FOV).
All stage axes have limited adjustment range, especially the Thorlabs stages. To deal with this, it is best to leave the stage base bolts and sample holder bolts loose and move the sample holder in x, y and z to roughly find your AOI. Once you are on or near your AOI, tighten the bolts and use the micrometers to center your image. One trick here is to get the z clamped first, then deal with x and y.
CMOS camera
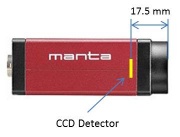
The microscope you will build does not have an eyepiece for direct visual observation. Instead, images will be captured with a CMOS camera[4]. Its monochrome (black and white) sensor contains a grid of 728x544 square pixels that measure 6.9 μm on a side. An adapter ring converts the C-mount thread on the camera to SM1.
Light sources
There are several different lights sources available in the lab that serve different purposes. In this part of Assignment 1, we will use a light source set up for trans-illumination of the sample, i.e. the sample will be illuminated from the opposite side as the objective lens. This type of illumination only works for samples that are mostly transparent. Later on, we will use a green LED to excite fluorescent microscopy samples. The green LED will be configured for epi-illumination - that is, it will illuminate the sample through the same objective lens that is used to image the sample. There will be more on this in Assignment 2.
For now, we will use a red LED to illuminate the sample. Any color can be used for bright-field transmitted light imaging. On one hand a blue LED yields a better bright-field resolution, however bright-field resolution is not usually critical for the samples we are interested in. On the other hand, a red LED allows simultaneous fluorescent and bright-field imaging, which you will be doing in later assignments. This can be quite useful when trying to bring a dim fluorescent sample into focus.
The LEDs we have in the lab produce light that is very divergent. You should use a condenser lens to collimate the light onto the sample. Since we want to collect as much of the LED light as possible, it's best to use a condenser lens with as small a focal length as possible. The 25 mm focal length lens works well as a condenser.
General advice
You can spend a huge amount of time wandering around the lab just collecting parts, so it makes sense to grab as many parts as possible in one trip.
Most of the materials you will need for each step are shown in an image gallery, including a part number and the manufacturer's name of of the part. When you come across a part name or number that is not-all-that-self-explanatory, remember that you grew up with the freakin' Internet in your pocket. Google is your friend. If you have a question about any of the components, the manufacturer's website can be very helpful. For example, if the procedure calls for an SPW602 spanner wrench and you have no idea what such a thing might look like, try googling the term: "thorlabs SPW602". You will find your virtual self just a click or two away from a handsome photo and detailed specifications.
Try to be nice to the components. It should never be necessary to force anything. When in doubt, ask before you break it rather than after.
Assemble the microscope
We recommend reproducing the general layout of the example microscope: it grants compactness and allows your device to be a stand-alone breadboard-transportable microscope. Even though you are building the bright-field imaging leg of your microscope, some of the following instructions will point you towards including key components for the fluorescence part of the microscope to save you time later.
Can you identify all the components of the block diagram? Try to think about the purpose of each additional component, and why it is laid out a particular way. Why are some sections of the light path open (strut-based structure, cage rods), while others are enclosed (Thorlabs lens tubes)?
Once you're satisfied that you understand the big picture, it's time to build your own!
Make sure to construct your microscope well. Mechanical stability will be crucial for the particle tracking experiments in the last part of the lab. The required stability specification will be achieved through good design and careful construction — not by indiscriminate over-tightening of screws. If you ever get stuck or are unsure along the way - just ask an instructor! We are here to help!
Assemble the base
A good place to start is to assemble what we’re going to call the “microscope base”.
This section of the microscope serves several functions: it holds the objective, it provides support when attached to the large 1.5” vertical post, it will also hold the dichroic mirror that you’ll use for fluorescence excitation in Assignment 2. Most of these parts can be found either on the counter or on the shelves above the west drawers.
- Notice that there are two subtly different cage cubes in this microscope assembly: a C6W and a C4W. The C4W only has small 4-40 tapped screw holes around its side, while the C6W has only two sides with 4-40 tapped holes, while the other two sides each have four larger holes for 30mm cage rods. You'll use one of each. Use the C4W as the bottom-most mirror holder. The C6W will stack on top of the C4W, and will later hold a dichroic mirror needed for fluorescence imaging.
- Attach four ER025 cage rods to the C4W. Ensure that each of the ER025 cage rods only have a single set screw (see figure below). If left in, the second set screw can interfere with the dichroic mirror mount that we'll add in Assignment 2.


- Secure the C6W cube onto the ER025 cage rods by tightening the four set screws adjacent to the rods.

- Use a round optic mount (B5C) to mount an aluminum mirror onto a kinematic platform (B4C). Use the clear plastic #4/40 screws affix the mirror without deforming nor scratching its surface. Attach the mirror/B5C assembly so that the mirror's reflective surface lies on exactly a diameter of the B4C mount. The mounting bracket should not stand in the way of the rotation of the kinematic platform. Take a look at the demo mount for an example of how everything fits together.
- Secure the mirror and B4C base into the C4W cage cube at a 45 degree angle. Use four black, plastic #4/40 screws to hold the cube optic mount (B4C) on the cage cube (C4W). Tighten the screws enough so that the B4C platform holds its adjustment, but can still be rotated.
- Attach a cage cube cover plate (B1C) to each cage cube (C4W and C6W) using 4-40 socket head cap screws. In Assignment 2 you will add a B4C platform holding a dichroic mirror to the C6W cube opposite the cover plate.
- Loosely screw in the 8-32 threads of a C1500 post clamp to the center of each B1C cage plate. DO NOT tighten the knob all the way! We will wait until mounting the microscope base to the 1.5" vertical post before clamping everything down.
- Finally, attach three ER2 2" cage rods to the C4W cube to complete the microscope base.

Secure the microscope base to the P14 post
Next, you might consider mounting your newly assembled microscope base to a sturdy P14 1.5” diameter post. Gather the following parts:
- Optical breadboard and P14 post located in the lower left cubby of the Instructor Cubbies
- Additional useful components located on the west cabinet
- Attach the PB1 base to the P14 post using a 1/4-20 screw.
- On a 1' x 2' x 1/2" optical breadboard, align the vertical Thorlabs P14 (1.5" diameter mounting post) with a breadboard hole that is12 positions from a short side and 3 positions from a long side. This allows enough free space on the breadboard to add things later.
- Secure the post base into the breadboard with 1/4-20 screws.
- Slide the two C1500 clamps from the microscope base over the 1.5" post, and tighten the screws in the order shown in the figure. This order will ensure that the base is leveled and secure, without putting unnecessary strain on the parts. The height of the top cube should be roughly 142 mm. Use the “handy scope thingamajig” to match your height to the example microscope’s.

Continue building out your microscope
Now that you're more familiar with the optomechanical building process, our microscope assembly instructions will become less and less detailed. Continue building out the rest of your scope using the example as your guide. Don't hesitate to ask an instructor if you are uncertain or have any questions. Keep in mind the following helpful tips as you build:
- Think about your block diagram and what distances between components are most important. In our microscope, we'll be using a 125 mm tube lens instead of the typical Nikon 200 mm one. Remember that for a 4f microscope system, you want your tube lens to be one focal length from the BFP of your objective, however, due to space constraints, the distance between your tube lens and objective might be slightly greater than 125 mm. What effect will this have on your microscope magnification? Do your best to get the distance right, but make sure there is enough room to insert and remove a SM1L05 lens tube (which will hold a barrier filter to be added next week).
- Use only three cage rods to connect the C4W cage cube to the CP02 cage plate, so you can easily take in and out the barrier filter (BF) that is needed for fluorescence microscopy. It’s good practice to always place two rails at the top, and leave the open slot at the bottom.
- Verify the focal length of all lenses you use in your scope. Previous classmates of yours do not always put things back where they belong. Shame on them. Assert your moral superiority: if you find an optic in the wrong box, identify it and either replace it in the correct box or label its current box correctly. (Ask an instructor if you can't find the right box. We have a stash of empty optics boxes near the wire spools behind you as you stand at the wet bench.)
- Check all your lenses and mirrors for cleanliness before you use them. You'll save yourself some troubleshooting time and effort down the road!
- Most of the time, lenses should be mounted in SM1L05 lens tubes. Use SM1RR retaining rings (and never an SM1T2, SM1V01, or SM1V05) to mount optics in lens tubes.
- Don't just drop the lens into the lens tube. Use lens paper to gently lower the lens into the tube.
- The lens will rest against a ridge inside of the tube, near the threaded end.
- Think about the orientation of the lens. Which side of the lens tube do you want the curved side to be on? the flat side?
- Don't touch the lens while you are putting it in. Fingerprints are the enemy of optics.
- Thread an SM1RR retaining ring into the tube and then tighten it with the red SPW602 spanner wrench. Ask an instructor for a demonstration if you don't understand.
- mounting lenses:
- You want every component to be fixed to the breadboard and well-supported. (The stability of your microscope will be important later in the semester). Secure large unsupported components to the breadboard with 0.5" posts (TR1) mounted in PH1 post holders connected to BA1 bases. See the photo below (note that the post holders in the photo are 1" taller than the one’s you’ll use. Please use a washer when securing the BA1 to the breadboard.
- Use adjustable mounting components in front of the CMOS camera so you can optimize and fine-tune the camera positing with respect to the imaging lens L2. Beware: never use an SM1T2 coupler without a locking ring — they are very difficult to remove if they are tightened against a lens tube or tube ring. Also put a quick-connect in your design such that the camera CMOS will end up 125 mm from the tube lens. Remember that the CMOS detector is recessed inside the opening of the camera.
Assemble the illuminator
This semester we have brand new, custom made, fancy shmancy LED mounts. These are made to interface with the Thorlabs cage system and mount to the P14 post so that it will be centered over your objective.
You might wonder why there is a strange star-shaped block attached to the back of the LED mount. This is a heatsink that will help keep your LEDs from overheating, preventing them from blowing out. While such a beefy heat sink may be overkill for the red LEDs we're using for now, the high power LEDs we'll use for fluorescence excitation need the extra protection.
- Light coming from an LED is very divergent. Use a 25 mm lens as a condenser to collimate the light, which will help to evenly illuminate your sample.
- You may noticed that in general, the shorter the focal length, the thicker the lens. With these very thick lenses (f = 25 mm or less), it's good practice to mount the lens using two retaining rings to clamp the lens in place, rather than letting the lens sit up again the SM1L05 lens tube.
- Thread an SM1RR retaining ring into a SM1L05 lens tube and use the SPW602 spanner wrench to drive it about 90% of the way down the tube.
- Place the 25 mm lens in the SM1L05 lens tube with the curved side facing the external threads of the tube.
- Thread a second SM1RR retaining ring into the lens tube and tighten it with the SPW602 spanner wrench.
- Screw the lens tube into an CP02 cage plate, and lock it into place.
- Secure the CP02 cage plate to the LED mount using 3 ER2 cage rods. Which side of the lens (curved or flat) should be closest to the LED?
- Mount the LED to the P14 post using a hex standoff (like you did for the microscope base) and a C1500 clamp.
- Connect the LED to power
- Turn the power supply on.
- Make sure the power supply is not enabled (green LED below the OUTPUT button is not lit).
- Use the righthand set of knobs to set the current and voltage
- Adjust the CH1/MASTER VOLTAGE knob so the display reads about 5 Volts.
- Adjust the CH1/MASTER CURRENT knob to so the display reads 0.1 Amps.
- IMPORTANT: Never set the CURRENT to a value greater than 0.5A, as this will burn out the LED.
- Pick up a red and a black cable from the cable rake in the corner of the lab. I like the ones with the flat horseshoe on one end and the alligator clip on the other.
- Connect the + (red) terminal of channel CH1 on the power supply to the red wire of the LED.
- Connect the - (black) terminal of channel CH1 on the power supply to the black wire of the LED.
| |
Double check your wiring before powering the LED. The LED can be damaged by excessive current, or by wiring it the wrong way. Limit the driving current to 0.5 A to protect the LED. |
- Press the OUTPUT button to enable the power supply and light the LED.
- Adjust the LED brightness using the power supply's CURRENT knob REMEMBERING NOT TO EXCEED 0.5A.
Once you're finished assembly...
Nice job! Take a minute to admire your work. You and this microscope will be spending a lot of time together this semester.
Now it's time to start testing and measuring things! If you need a break and won't be completing Part 4 of Assignment 1 immediately, read the instructions for microscope storage at the end of Part 4.
Back to Assignment 1
References
- ↑ Hooke, R. Micrographia: or Some Physiological Descriptions of Minute Bodies made by Magnifying Glasses with Observations and Inquiries Thereupon London:Jo. Martyn, and Ja. Allestry, Printers to the Royal Society; 1665
- ↑ See, for example: McClintock, B. The origin and behavior of mutable loci in maize. PNAS. 1950; 36:344-355. and Endersby, Jim. A Guinea Pig's History of Biology. Cambridge, Massachusetts: Harvard University Press; 2007.
- ↑ Precision Newport X/Y/Z stages
- ↑ Allied Manta G040
- Overview
- Part 1: Pre-lab questions
- Part 2: Optics bootcamp
- Part 3: Build a microscope
- Part 4: Measure stuff
Back to 20.309 Main Page