Difference between revisions of "20.109(F09): Mod 3 Day 5 Battery testing"
(→Introduction) |
(→Introduction) |
||
| Line 3: | Line 3: | ||
=<center>Battery Testing</center>= | =<center>Battery Testing</center>= | ||
==Introduction== | ==Introduction== | ||
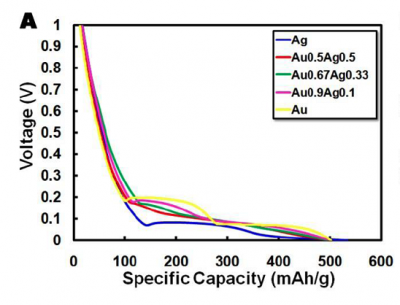
| − | [[Image:Charge-discharge.png|thumb|left|400px]] | + | [[Image:Charge-discharge.png|thumb|left|400px]]Take a look at your cell phone battery. If you have a typical LiCoO2 battery you will notice that it has a voltage (3.7 V) and a capacity (e.g. 710mAh for the Motorola MOTORAZR V3r battery). We know that LiCoO2 has a theoretical capacity of 130 mAh/g, so it's possible to back calculate the amount of active material as 5.46 g. The way Motorola determined the capacity and voltage for their cell phone battery is by testing them on a Galvanostat. This machine applies either positive or negative current in order to charge or discharge a battery, just as you will be doing in lab today. By attaching the batteries to a Galvanostat and applying a constant current (1100 mA), the battery will charge in a given time interval (in our case 10 hours). By applying a negative current of 1100 mA it's possible to discharge the battery in that same amount of time. The discharge graph, such as the one shown here, plots voltage vs percentage of capacity discharged. The flatter the curve the better since this means voltage remains constant as the battery is used up. As an example, |
| − | Once we know the capacity of the battery material, it's possible to | + | The theoretical capacity of our AuNW’s, based on the 15:4 Li:Au ratio, is 510mAh/g. The theoretical capacity is determined The value of 50mA/g is a current “loading factor” for how much current to use for a 10h discharge (multiply the capacity of ~500mAh/g*10h = 50mA/g). is 510mAh/g. The value of 50mA/g is a current “loading factor” for how much current to use for a 10h discharge (multiply the capacity of ~500mAh/g*10h = 50mA/g). |
| + | |||
| + | Once we know the capacity of the battery material, it's also possible to calculate the charge rate. We will be testing everything through 2 cycles of 10 hours each; this means that when we charge the battery it should take one hour, and it should discharge in one hour. Since every electrode weighs a little different, the charge/discharge rate will be slightly different. As an example, if I have an electrode of FePO4•H2O that has 1 mg of active material, then in order to discharge it in one hour I have to put a negative current of -143 uA. | ||
<br><center>1 mg x143 mAh/g x 1 g/1000 mg x 1/1hr = 143uA</center><br> | <br><center>1 mg x143 mAh/g x 1 g/1000 mg x 1/1hr = 143uA</center><br> | ||
| − | The electrodes that were prepared during last session were placed in a vacuum oven and dried at 100°C for four hours. They were then moved into a glove box and crimped into a battery. Today instruction will be given on how to | + | |
| + | ==Protocol== | ||
| + | The electrodes that were prepared during last session were placed in a vacuum oven and dried at 100°C for four hours. They were then moved into a glove box and crimped into a battery, with the gold nanowires serving as the batteries cathode. Today instruction will be given on how to measure the charge/discharge on the Galvanostat. | ||
Revision as of 18:49, 27 November 2009
Battery Testing
Introduction
Take a look at your cell phone battery. If you have a typical LiCoO2 battery you will notice that it has a voltage (3.7 V) and a capacity (e.g. 710mAh for the Motorola MOTORAZR V3r battery). We know that LiCoO2 has a theoretical capacity of 130 mAh/g, so it's possible to back calculate the amount of active material as 5.46 g. The way Motorola determined the capacity and voltage for their cell phone battery is by testing them on a Galvanostat. This machine applies either positive or negative current in order to charge or discharge a battery, just as you will be doing in lab today. By attaching the batteries to a Galvanostat and applying a constant current (1100 mA), the battery will charge in a given time interval (in our case 10 hours). By applying a negative current of 1100 mA it's possible to discharge the battery in that same amount of time. The discharge graph, such as the one shown here, plots voltage vs percentage of capacity discharged. The flatter the curve the better since this means voltage remains constant as the battery is used up. As an example,The theoretical capacity of our AuNW’s, based on the 15:4 Li:Au ratio, is 510mAh/g. The theoretical capacity is determined The value of 50mA/g is a current “loading factor” for how much current to use for a 10h discharge (multiply the capacity of ~500mAh/g*10h = 50mA/g). is 510mAh/g. The value of 50mA/g is a current “loading factor” for how much current to use for a 10h discharge (multiply the capacity of ~500mAh/g*10h = 50mA/g).
Once we know the capacity of the battery material, it's also possible to calculate the charge rate. We will be testing everything through 2 cycles of 10 hours each; this means that when we charge the battery it should take one hour, and it should discharge in one hour. Since every electrode weighs a little different, the charge/discharge rate will be slightly different. As an example, if I have an electrode of FePO4•H2O that has 1 mg of active material, then in order to discharge it in one hour I have to put a negative current of -143 uA.