Difference between revisions of "20.109(S08):Module 1"
(→References) |
|||
| Line 62: | Line 62: | ||
#'''Homologous recombination as a mechanism of carcinogenesis'''<br>'' Biochim Biophys Acta'' 21 March 2001<br> Bishop AJ and Schiestl RH<br> [http://dx.doi.org/10.1016/S0304-419X(01)00018-X URL] | #'''Homologous recombination as a mechanism of carcinogenesis'''<br>'' Biochim Biophys Acta'' 21 March 2001<br> Bishop AJ and Schiestl RH<br> [http://dx.doi.org/10.1016/S0304-419X(01)00018-X URL] | ||
#'''Rad51-deficient vertebrate cells accumulate chromosomal breaks prior to cell death'''<br>'' EMBO J'' 15 January 1998<br> E Sonoda, M S Sasaki, J M Buerstedde, O Bezzubova, A Shinohara, H Ogawa, M Takata, Y Yamaguchi-Iwai, and S Takeda<br> [http://www.pubmedcentral.gov/picrender.fcgi?artid=1170409&blobtype=pdf PDF reprint] | #'''Rad51-deficient vertebrate cells accumulate chromosomal breaks prior to cell death'''<br>'' EMBO J'' 15 January 1998<br> E Sonoda, M S Sasaki, J M Buerstedde, O Bezzubova, A Shinohara, H Ogawa, M Takata, Y Yamaguchi-Iwai, and S Takeda<br> [http://www.pubmedcentral.gov/picrender.fcgi?artid=1170409&blobtype=pdf PDF reprint] | ||
| + | |||
| + | [[20.109(S08): TA notes for module 1| TA notes, mod 1]] | ||
Revision as of 18:55, 27 December 2007
Module 1
Instructors: Bevin Engelward and Agi Stachowiak
TA: David Weingeist
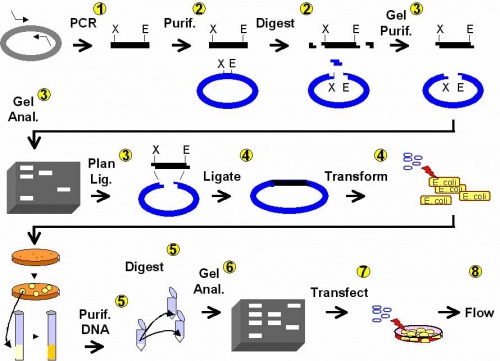
In this experimental module you will modify the gene for EGFP (Enhanced Green Fluorescent Protein) to truncate the protein it encodes. Cells expressing the full-length protein glow green when exposed to light of the appropriate wavelength. You will be designing and then creating an expression vector to delete the first 32 amino acids of EGFP. Cells transfected with your expression vector should not glow green, a prediction you will test. You will also test whether this N-terminally truncated EGFP can recombine with a C-terminally truncated version to regenerate full length EGFP in vivo. Finally, you will have the opportunity to suggest changes to the experimental protocol that will increase the frequency of green cells in which there has been an inter-plasmid recombination event. We will then choose a few variables to test on the final day of the experiment.
Lab handouts
Day 1: DNA engineering using PCR (you will also need weblinks, below)
Day 3: Agarose gel electrophoresis
Day 4: DNA ligation and bacterial transformation
Day 5: Examine candidate clones
Day 6: Restriction map and tissue culture
Module 1 lab report schedule and guidelines
DNA engineering web links
Engelward lab resources: https://web.mit.edu/bevin/www/UltiMouse/
pCX-EGFP plasmid map: https://web.mit.edu/bevin/www/UltiMouse/pCX-EGFP.pdf File:Macintosh HD-Users-nkuldell-Desktop-pCX-EGFP.doc
ORF finder: http://www.ncbi.nlm.nih.gov/gorf/gorf.html
NCBI: http://www.ncbi.nlm.nih.gov/
Cybergene: http://www.cybergene.se/primer.html
New England Biolabs: http://www.neb.com/nebecomm/default.asp
References
Note: PDF reprints are provided below within the context of fair use. Please obtain copies from the publisher if appropriate.
- DNA double-strand break repair: From mechanistic understanding to cancer treatment
DNA Repair 2007
Thomas Helleday, Justin Lo, Dik C. van Gent, Bevin P. Engelward
URL - Homologous recombination as a mechanism of carcinogenesis
Biochim Biophys Acta 21 March 2001
Bishop AJ and Schiestl RH
URL - Rad51-deficient vertebrate cells accumulate chromosomal breaks prior to cell death
EMBO J 15 January 1998
E Sonoda, M S Sasaki, J M Buerstedde, O Bezzubova, A Shinohara, H Ogawa, M Takata, Y Yamaguchi-Iwai, and S Takeda
PDF reprint