Difference between revisions of "20.109(S08):Characterize protein expression (Day5)"
(→Introduction) |
MAXINE JONAS (Talk | contribs) m (68 revisions: Transfer 20.109(S08) to HostGator) |
||
| (11 intermediate revisions by 2 users not shown) | |||
| Line 5: | Line 5: | ||
Last time you used the lactose-analogue IPTG to induce expression of inverse pericam in BL21(DE3) bacteria. Today you will isolate IPC from the bacteria, and you will begin characterizing your wild-type and mutant proteins. | Last time you used the lactose-analogue IPTG to induce expression of inverse pericam in BL21(DE3) bacteria. Today you will isolate IPC from the bacteria, and you will begin characterizing your wild-type and mutant proteins. | ||
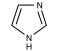
| − | We can take several measures to ensure that a high quantity of plasmid-encoded protein is produced by our bacteria, such as using a high-copy plasmid. However, the bacteria in which we grow the protein clearly need to produce other proteins merely to survive. The bacterial expression vector we are using [https://catalog.invitrogen.com/index.cfm?fuseaction=viewCatalog.viewProductDetails&productDescription=615 (pRSET)] contains six Histidine residues downstream of a bacterial promoter and in-frame with a start codon. Our resultant protein is therefore marked by the presence of these residues, or His-tagged. Histidine has several interesting properties, notably its near-neutral pKa, and His-rich peptides are promiscuous binders, particularly to metals. | + | We can take several measures to ensure that a high quantity of plasmid-encoded protein is produced by our bacteria, such as using a high-copy plasmid. However, the bacteria in which we grow the protein clearly need to produce other proteins merely to survive. The bacterial expression vector we are using [https://catalog.invitrogen.com/index.cfm?fuseaction=viewCatalog.viewProductDetails&productDescription=615 (pRSET)] contains six Histidine residues downstream of a bacterial promoter and in-frame with a start codon. Our resultant protein is therefore marked by the presence of these residues, or His-tagged. Histidine has several interesting properties, notably its near-neutral pKa, and His-rich peptides are promiscuous binders, particularly to metals. (For example, histidine side chains help coordinate iron molecules in hemoglobin.) |
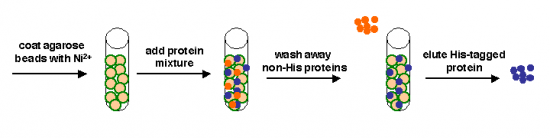
[[Image:20.109_Ni-Ag-Schematic.png|thumb|550px|right|'''Affinity separation process''' Green represents Nickel, blue the (His-tagged) protein of interest, and orange the other proteins in the cell extract.]] | [[Image:20.109_Ni-Ag-Schematic.png|thumb|550px|right|'''Affinity separation process''' Green represents Nickel, blue the (His-tagged) protein of interest, and orange the other proteins in the cell extract.]] | ||
| Line 14: | Line 14: | ||
| − | Prior to purifying our protein, we will lyse the bacteria, and run whole bacterial extracts on a protein gel. This procedure is called SDS-PAGE, for sodium docecyl sulfate-polyacrylamide gel electrophoresis. SDS is an ionic surfactant (or detergent), which denatures the proteins and coats them with a negative charge. Since denatured proteins are linear, they will move through the gel at a speed inversely proportional to their molecular weight, just like DNA on agarose gels. (Non-denatured proteins run according to their molecular weight, shape, and charge.) As we did with DNA gels, we will run a reference ladder containing proteins of known molecular weight and amount. When running | + | Prior to purifying our protein, we will lyse the bacteria, and run whole bacterial extracts on a protein gel. This procedure is called SDS-PAGE, for sodium docecyl sulfate-polyacrylamide gel electrophoresis. SDS is an ionic surfactant (or detergent), which denatures the proteins and coats them with a negative charge. Since denatured proteins are linear, they will move through the gel at a speed inversely proportional to their molecular weight, just like DNA on agarose gels. (Non-denatured proteins run according to their molecular weight, shape, and charge.) As we did with DNA gels, we will run a reference ladder containing proteins of known molecular weight and amount. When running ���IPTG and +IPTG samples side-by-side, you should see the emergence of a protein band at the expected molecular weight for inverse pericam, which may be very faint or non-existent in the control sample, but bright and thick in the induced sample. To visualize all the proteins released by the bacteria, you will stain the gels with Coomassie Brilliant Blue (actually, a variant called BioSafe Coomassie). This is a non-specific stain for all proteins. In a technique called Western Blotting, SDS-PAGE is combined with the use of antibodies to preferentially stain a single protein. |
| − | After purifying inverse pericam from your bacterial lysates, you will measure the protein concentration by the Bradford colorimetric assay, named after the scientist who first [http://www.ncbi.nlm.nih.gov/pubmed/942051 published it]. The dye used in this assay is the same one you will use to stain your protein gels | + | After purifying inverse pericam from your bacterial lysates, you will measure the protein concentration by the Bradford colorimetric assay, named after the scientist who first [http://www.ncbi.nlm.nih.gov/pubmed/942051 published it]. The dye used in this assay is the same one you will use to stain your protein gels ��� Coomassie. In acidic solution, Coomassie normally has an absorbance peak at ~ 465 nm (blue light), but this peak is shifted to 595 nm (orange light) upon binding to protein. Protein binding occurs primarily via arginine, as well as other basic and aromatic residues, as described [http://www.ncbi.nlm.nih.gov/pubmed/4096375 here]. The concentration of protein present in a sample is thus proportional to the 595 nm absorbance peak, and its absolute value can be determined using a standard curve of reference protein. We do not have a sample of inverse pericam with a known quantity of protein, so today we will use BSA as a reference protein. Because the compositions of IPC and BSA with respect to arginine may vary, this assay will really only give the relative concentrations of your protein samples, and the absolute concentrations will have an associated error. |
==Protocols== | ==Protocols== | ||
| Line 31: | Line 31: | ||
===Part 2: SDS-PAGE of protein extracts=== | ===Part 2: SDS-PAGE of protein extracts=== | ||
| − | #Last time you measured the amount of cells in each of your samples. (If you ran cultures overnight, the teaching faculty measured the +IPTG samples for you and | + | #Last time you measured the amount of cells in each of your samples. (If you ran cultures overnight, the teaching faculty measured the +IPTG samples for you and posted the results.) Look back at your measurements, and find the sample with the lowest cell concentration. Set aside 15 μL of this sample for PAGE analysis in an eppendorf. |
#For your other five samples, you should take the amount of bacterial lysate corresponding to the same number of cells as the lowest concentration sample. For example, if the OD<sub>600</sub> of your WT -IPTG sample was 0.05, and the OD<sub>600</sub> of your WT +IPTG sample was 0.30, you would take 15 μL of the -IPTG, but only 2.5 μL of the +IPTG sample. | #For your other five samples, you should take the amount of bacterial lysate corresponding to the same number of cells as the lowest concentration sample. For example, if the OD<sub>600</sub> of your WT -IPTG sample was 0.05, and the OD<sub>600</sub> of your WT +IPTG sample was 0.30, you would take 15 μL of the -IPTG, but only 2.5 μL of the +IPTG sample. | ||
#Next, add enough water so the each sample has 15 μL of liquid in it. You might use the table below to guide your work. | #Next, add enough water so the each sample has 15 μL of liquid in it. You might use the table below to guide your work. | ||
| Line 93: | Line 93: | ||
</center> | </center> | ||
| − | #Now add 15 | + | #Now add 15 ��L of 2X sample buffer to 15 ��L of each of your diluted lysates. Also retrieve 15 ��L samples of MW markers from the teaching faculty. |
#*The pre-stained marker (lane 0) will be used to track the progress of the gel. | #*The pre-stained marker (lane 0) will be used to track the progress of the gel. | ||
#*The unstained marker (lane 7) contains a known amount of protein per band, and will be used to estimate the gross protein contents of your samples. | #*The unstained marker (lane 7) contains a known amount of protein per band, and will be used to estimate the gross protein contents of your samples. | ||
| − | #Boil all eight eppendorfs for 5 minutes in the water bath. | + | #Boil all eight eppendorfs for 5 minutes in the water bath that is in the fume hood. |
#You will be shown by the teaching faculty how to load your samples into the gel. You might load your samples according to the table above. | #You will be shown by the teaching faculty how to load your samples into the gel. You might load your samples according to the table above. | ||
#Note the starting and stopping time of electrophoresis, which will be initiated by the teaching faculty at 200 V, and run for 30-45 minutes. | #Note the starting and stopping time of electrophoresis, which will be initiated by the teaching faculty at 200 V, and run for 30-45 minutes. | ||
| − | #Pry apart the plates using a spatula, and transfer your gel to a staining box. Add Coomassie | + | #Pry apart the plates using a spatula, and carefully transfer your gel to a staining box. Add 200 mL of deonized water and rinse the gel for 5 min. |
| − | # | + | #Repeat the rinse two more times with fresh water (200 mL and 5 min incubation each time). |
| − | #Replace the | + | #Add 50 mL of BioSafe Coomassie, and incubate for at least 1 hour. |
| − | #Tomorrow, the teaching staff will transfer each gel to water, then photograph them and post the results to the wiki. You will have a chance to physically observe your gels next time. | + | #Empty the staining solution into the waste container in the fume hood - careful not to lose your gel! |
| + | #Add 200 mL of water to your stained gel. Replace with fresh water just before leaving the lab if you have a chance. | ||
| + | #Tomorrow, the teaching staff will transfer each gel to fresh water, then photograph them and post the results to the wiki. You will have a chance to physically observe your gels next time. | ||
===Part 3: Protein purification=== | ===Part 3: Protein purification=== | ||
| − | You will process three samples (the three +IPTG extracts) according to the following procedure. You should either time your spins with another group, or | + | You will process three samples (the three +IPTG extracts) according to the following procedure. You should either time your spins with another group, or balance your tubes with 3-way symmetry. '''Keep all buffers on ice when not in use.''' <big>'''All spins should be performed at 1000 rcf for 1 min.'''</big> |
#The following buffers have been prepared for you: | #The following buffers have been prepared for you: | ||
| Line 114: | Line 116: | ||
#*Elute Buffer (0.5 M NaCl, 10 mM Tris-HCl, 1 M imidazole, pH 7.9) | #*Elute Buffer (0.5 M NaCl, 10 mM Tris-HCl, 1 M imidazole, pH 7.9) | ||
#*All but the Charge Buffer contain protease inhibitors to help keep your protein intact. | #*All but the Charge Buffer contain protease inhibitors to help keep your protein intact. | ||
| − | #Gently rock the nickel-agarose resin to fully resuspend it, then distribute 400 | + | #Gently rock the nickel-agarose resin to fully resuspend it, then distribute 400 ��L of slurry to each of three tubes. |
# Label each tube as wild-type or mutant, then spin for 1 min. at low speed. | # Label each tube as wild-type or mutant, then spin for 1 min. at low speed. | ||
| − | #Remove the 200 | + | #Remove the 200 ��L of supernatant from the resin and add it to your waste collection tube. The damp, semi-solid resin left behind should be ~ 200 ��L ���tall.��� |
| − | #First you must rinse the resin. Add 400 | + | #First you must rinse the resin. Add 400 ��L of sterile DI water to each tube. Place on the nutator for 15-60 seconds to mix, or simply invert the tube several times. Flick the tube to compete resuspension of the resin if necessary. |
| − | #Spin for 1 min., then pipet off and discard the entire supernatant (400 | + | #Spin for 1 min., then pipet off and discard the entire supernatant (400 ��L). |
#Repeat steps 5 and 6 for the following buffers | #Repeat steps 5 and 6 for the following buffers | ||
#*a second wash with DI water | #*a second wash with DI water | ||
| − | #*3 washes with Charge Buffer, 400 | + | #*3 washes with Charge Buffer, 400 ��L each time |
| − | #*2 washes with Binding Buffer, 400 | + | #*2 washes with Binding Buffer, 400 ��L each time |
| − | #Add your entire cell extract to the resin (~550-600 | + | #Add your entire cell extract to the resin (~550-600 ��L). Be sure to add each sample to the appropriately labeled tube! |
#Invert to mix the three samples as usual, then place on the nutator for 5 min. Spin and discard supernatants as before. | #Invert to mix the three samples as usual, then place on the nutator for 5 min. Spin and discard supernatants as before. | ||
#Now you will again repeat steps 5 and 6, to wash away contaminants: | #Now you will again repeat steps 5 and 6, to wash away contaminants: | ||
| − | #*3 washes with Binding Buffer, 600 | + | #*3 washes with Binding Buffer, 600 ��L each time |
| − | #*2 washes with Wash Buffer, 600 | + | #*2 washes with Wash Buffer, 600 ��L each time |
| − | #Finally, you will collect your protein. Add 500 | + | #Finally, you will collect your protein. Add 500 ��L of Elute Buffer, resuspend and spin as usual. Do not throw away the supernatant! Instead, transfer it to a fresh eppendorf tube, labeled ���pure IPC X#Z��� or ���pure WT.��� |
| − | #Do not throw away the resin yet either! Instead, add another 500 | + | #Do not throw away the resin yet either! Instead, add another 500 ��L of Elute Buffer, and repeat the step above. Add the second supernatant to the first. |
| − | #Immediately after eluting your protein, transfer 10 | + | #Immediately after eluting your protein, transfer 10 ��L of it to a clean eppendorf tube (for assaying protein concentrations), and add a 1:100 dilution of BSA to the remaining protein (10 ��L of BSA for ~ 1 mL of protein). |
===Part 4: Protein Concentration=== | ===Part 4: Protein Concentration=== | ||
| − | #Prepare | + | #Prepare 12 mL Bradford reagent from the 5x concentrated stock by adding water. |
#Obtain BSA standards from the teaching faculty. The standards were prepared in elution buffer, since imidazole has some absorbance at 595 nm. | #Obtain BSA standards from the teaching faculty. The standards were prepared in elution buffer, since imidazole has some absorbance at 595 nm. | ||
#*Each tube already contains exactly 10 μL of standard (or plain elution buffer, for your blank solution). | #*Each tube already contains exactly 10 μL of standard (or plain elution buffer, for your blank solution). | ||
| Line 181: | Line 183: | ||
==For next time== | ==For next time== | ||
| + | #Next time in lab, only two groups at a time will come to work. Please [[Talk:20.109%28S08%29:Assay_protein_behavior_%28Day6%29 |sign up]] for a time slot on the "talk" page of Day 6. | ||
#Prepare a table listing the protein concentrations of the samples you assayed today, and write a short caption for it. (This will feed into Part 2 of the portfolio.) | #Prepare a table listing the protein concentrations of the samples you assayed today, and write a short caption for it. (This will feed into Part 2 of the portfolio.) | ||
#Explain why the Bradford reagent turns from brown to blue, and not, say, from blue to red. | #Explain why the Bradford reagent turns from brown to blue, and not, say, from blue to red. | ||
#If we wanted to perform a Western blot, what kind of antibody might we use to select for inverse pericam from a complex protein mixture? Name two possibilities. | #If we wanted to perform a Western blot, what kind of antibody might we use to select for inverse pericam from a complex protein mixture? Name two possibilities. | ||
Latest revision as of 16:04, 15 June 2015
Contents
Introduction
Last time you used the lactose-analogue IPTG to induce expression of inverse pericam in BL21(DE3) bacteria. Today you will isolate IPC from the bacteria, and you will begin characterizing your wild-type and mutant proteins.
We can take several measures to ensure that a high quantity of plasmid-encoded protein is produced by our bacteria, such as using a high-copy plasmid. However, the bacteria in which we grow the protein clearly need to produce other proteins merely to survive. The bacterial expression vector we are using (pRSET) contains six Histidine residues downstream of a bacterial promoter and in-frame with a start codon. Our resultant protein is therefore marked by the presence of these residues, or His-tagged. Histidine has several interesting properties, notably its near-neutral pKa, and His-rich peptides are promiscuous binders, particularly to metals. (For example, histidine side chains help coordinate iron molecules in hemoglobin.)
Today we will use a Nickel-agarose resin to separate our protein of interest from the other proteins present in the bacteria. The His-tagged protein will preferentially bind to the Nickel-coated beads, while irrelevant proteins can be washed away. Finally, a high concentration of imidazole (which is the side chain of histidine) can be used to elute the His-tagged inverse pericam by competition. Due to the inherent fragility of IPC, we will add several components to our protein extraction and purification reagents: bovine serum albumin (BSA), which is a protein stabilizer, and a cocktail of protease inhibitors.
Prior to purifying our protein, we will lyse the bacteria, and run whole bacterial extracts on a protein gel. This procedure is called SDS-PAGE, for sodium docecyl sulfate-polyacrylamide gel electrophoresis. SDS is an ionic surfactant (or detergent), which denatures the proteins and coats them with a negative charge. Since denatured proteins are linear, they will move through the gel at a speed inversely proportional to their molecular weight, just like DNA on agarose gels. (Non-denatured proteins run according to their molecular weight, shape, and charge.) As we did with DNA gels, we will run a reference ladder containing proteins of known molecular weight and amount. When running ���IPTG and +IPTG samples side-by-side, you should see the emergence of a protein band at the expected molecular weight for inverse pericam, which may be very faint or non-existent in the control sample, but bright and thick in the induced sample. To visualize all the proteins released by the bacteria, you will stain the gels with Coomassie Brilliant Blue (actually, a variant called BioSafe Coomassie). This is a non-specific stain for all proteins. In a technique called Western Blotting, SDS-PAGE is combined with the use of antibodies to preferentially stain a single protein.
After purifying inverse pericam from your bacterial lysates, you will measure the protein concentration by the Bradford colorimetric assay, named after the scientist who first published it. The dye used in this assay is the same one you will use to stain your protein gels ��� Coomassie. In acidic solution, Coomassie normally has an absorbance peak at ~ 465 nm (blue light), but this peak is shifted to 595 nm (orange light) upon binding to protein. Protein binding occurs primarily via arginine, as well as other basic and aromatic residues, as described here. The concentration of protein present in a sample is thus proportional to the 595 nm absorbance peak, and its absolute value can be determined using a standard curve of reference protein. We do not have a sample of inverse pericam with a known quantity of protein, so today we will use BSA as a reference protein. Because the compositions of IPC and BSA with respect to arginine may vary, this assay will really only give the relative concentrations of your protein samples, and the absolute concentrations will have an associated error.
Protocols
Part 1: Lysis of cells producing wild-type and mutant IPC
- You will be given an aliquot of room temperature BPER (bacterial protein extraction reagent), which also contains 0.1% bovine serum albumin (BSA, a stabilizer), and a protease inhibitor cocktail to guard against protein degradation. When you are ready to begin, add 1:1000 of cold enzyme mixture (obtained from teaching staff) to the BPER solution.
- Per cell pellet (6 total), add the appropriate volume of enzyme-containing BPER and resuspend by pipetting until the solution is relatively homogeneous.
- Resuspend -IPTG samples in 300 μL, and +IPTG samples in 600 μL - do you remember why?
- Vortex for 30-60 seconds.
- Incubate the solutions (at room temperature) for 3 min.
- Finally, spin for 3 min. at maximum speed and transfer supernatants to fresh tubes.
Part 2: SDS-PAGE of protein extracts
- Last time you measured the amount of cells in each of your samples. (If you ran cultures overnight, the teaching faculty measured the +IPTG samples for you and posted the results.) Look back at your measurements, and find the sample with the lowest cell concentration. Set aside 15 μL of this sample for PAGE analysis in an eppendorf.
- For your other five samples, you should take the amount of bacterial lysate corresponding to the same number of cells as the lowest concentration sample. For example, if the OD600 of your WT -IPTG sample was 0.05, and the OD600 of your WT +IPTG sample was 0.30, you would take 15 μL of the -IPTG, but only 2.5 μL of the +IPTG sample.
- Next, add enough water so the each sample has 15 μL of liquid in it. You might use the table below to guide your work.
| Sample/Lane # | Sample Name | OD600 | Sample Volume | Water Volume |
|---|---|---|---|---|
| 0 | pre-stained ladder | --- | --- | --- |
| 1 | ||||
| 2 | ||||
| 3 | ||||
| 4 | ||||
| 5 | ||||
| 6 | ||||
| 7 | unstained ladder | --- | --- | --- |
- Now add 15 ��L of 2X sample buffer to 15 ��L of each of your diluted lysates. Also retrieve 15 ��L samples of MW markers from the teaching faculty.
- The pre-stained marker (lane 0) will be used to track the progress of the gel.
- The unstained marker (lane 7) contains a known amount of protein per band, and will be used to estimate the gross protein contents of your samples.
- Boil all eight eppendorfs for 5 minutes in the water bath that is in the fume hood.
- You will be shown by the teaching faculty how to load your samples into the gel. You might load your samples according to the table above.
- Note the starting and stopping time of electrophoresis, which will be initiated by the teaching faculty at 200 V, and run for 30-45 minutes.
- Pry apart the plates using a spatula, and carefully transfer your gel to a staining box. Add 200 mL of deonized water and rinse the gel for 5 min.
- Repeat the rinse two more times with fresh water (200 mL and 5 min incubation each time).
- Add 50 mL of BioSafe Coomassie, and incubate for at least 1 hour.
- Empty the staining solution into the waste container in the fume hood - careful not to lose your gel!
- Add 200 mL of water to your stained gel. Replace with fresh water just before leaving the lab if you have a chance.
- Tomorrow, the teaching staff will transfer each gel to fresh water, then photograph them and post the results to the wiki. You will have a chance to physically observe your gels next time.
Part 3: Protein purification
You will process three samples (the three +IPTG extracts) according to the following procedure. You should either time your spins with another group, or balance your tubes with 3-way symmetry. Keep all buffers on ice when not in use. All spins should be performed at 1000 rcf for 1 min.
- The following buffers have been prepared for you:
- Charge Buffer (50 mM NiSO4)
- Binding Buffer (0.5 M NaCl, 20 mM Tris-HCl, 5 mM imidazole, pH 7.9)
- Wash Buffer (0.5 M NaCl, 20 mM Tris-HCl, 60 mM imidazole, pH 7.9)
- Elute Buffer (0.5 M NaCl, 10 mM Tris-HCl, 1 M imidazole, pH 7.9)
- All but the Charge Buffer contain protease inhibitors to help keep your protein intact.
- Gently rock the nickel-agarose resin to fully resuspend it, then distribute 400 ��L of slurry to each of three tubes.
- Label each tube as wild-type or mutant, then spin for 1 min. at low speed.
- Remove the 200 ��L of supernatant from the resin and add it to your waste collection tube. The damp, semi-solid resin left behind should be ~ 200 ��L ���tall.���
- First you must rinse the resin. Add 400 ��L of sterile DI water to each tube. Place on the nutator for 15-60 seconds to mix, or simply invert the tube several times. Flick the tube to compete resuspension of the resin if necessary.
- Spin for 1 min., then pipet off and discard the entire supernatant (400 ��L).
- Repeat steps 5 and 6 for the following buffers
- a second wash with DI water
- 3 washes with Charge Buffer, 400 ��L each time
- 2 washes with Binding Buffer, 400 ��L each time
- Add your entire cell extract to the resin (~550-600 ��L). Be sure to add each sample to the appropriately labeled tube!
- Invert to mix the three samples as usual, then place on the nutator for 5 min. Spin and discard supernatants as before.
- Now you will again repeat steps 5 and 6, to wash away contaminants:
- 3 washes with Binding Buffer, 600 ��L each time
- 2 washes with Wash Buffer, 600 ��L each time
- Finally, you will collect your protein. Add 500 ��L of Elute Buffer, resuspend and spin as usual. Do not throw away the supernatant! Instead, transfer it to a fresh eppendorf tube, labeled ���pure IPC X#Z��� or ���pure WT.���
- Do not throw away the resin yet either! Instead, add another 500 ��L of Elute Buffer, and repeat the step above. Add the second supernatant to the first.
- Immediately after eluting your protein, transfer 10 ��L of it to a clean eppendorf tube (for assaying protein concentrations), and add a 1:100 dilution of BSA to the remaining protein (10 ��L of BSA for ~ 1 mL of protein).
Part 4: Protein Concentration
- Prepare 12 mL Bradford reagent from the 5x concentrated stock by adding water.
- Obtain BSA standards from the teaching faculty. The standards were prepared in elution buffer, since imidazole has some absorbance at 595 nm.
- Each tube already contains exactly 10 μL of standard (or plain elution buffer, for your blank solution).
- Add 1 mL of Bradford reagent to each standard, as well as to your three unknown protein samples. Incubate 10-20 min at room temperature.
- Measure the absorbance of each sample at 595 nm. Work as quickly as you can, because the absorbance will continue to slowly change over time. To get a sense of the error incurred due to the ongoing reaction, measure your blank sample both at the beginning and at the end of your run.
| Sample (mg/mL) |
A595 | Sample | A595 |
|---|---|---|---|
| BSA 0.1 | Blank - start | ||
| BSA 0.2 | WT IPC | ||
| BSA 0.4 | Mutant 1 | ||
| BSA 0.6 | Mutant 2 | ||
| BSA 0.8 | Blank - end | ||
| BSA 1.0 | ----------- | ----------- |
For next time
- Next time in lab, only two groups at a time will come to work. Please sign up for a time slot on the "talk" page of Day 6.
- Prepare a table listing the protein concentrations of the samples you assayed today, and write a short caption for it. (This will feed into Part 2 of the portfolio.)
- Explain why the Bradford reagent turns from brown to blue, and not, say, from blue to red.
- If we wanted to perform a Western blot, what kind of antibody might we use to select for inverse pericam from a complex protein mixture? Name two possibilities.